Description
Gravity Blood Set, Non-Vented
The Gravity Blood Set, Non-Vented is a single-use IV administration set designed for gravity delivery of whole blood and compatible blood components in acute care, perioperative, emergency, and infusion settings. Built specifically for blood administration, it incorporates an integral blood filter to help reduce the risk of transfusing clots and aggregates while maintaining a clear, controlled flow path from container to patient. The non-vented spike is intended for use with collapsible blood bags and other flexible containers where air venting is not required.
Key Features
- Non-vented spike for flexible blood bags, supporting a closed system approach without introducing external air.
- Integral blood filter (typically 170–260 micron) within the drip chamber to screen macro-aggregates commonly associated with blood products.
- Gravity-controlled delivery with a roller clamp for simple, responsive flow adjustment.
- Clear, kink-resistant tubing to support visual monitoring for air, flow, or discoloration during transfusion.
- Standard luer-type patient connection designed to interface with common IV access devices and extension sets.
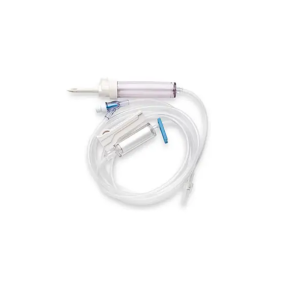
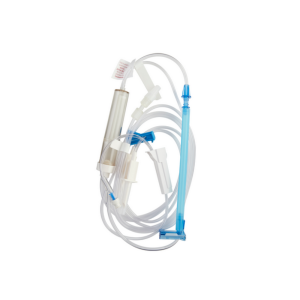
Major Components
| Component | Purpose |
|---|---|
| Non-vented piercing spike | Accesses the blood container without an air inlet; best suited to collapsible bags. |
| Drip chamber with blood filter | Allows drip visualization and filtration of clots/aggregates before entry into the tubing. |
| Roller clamp | Provides manual regulation of flow during gravity administration. |
| Administration tubing | Transports filtered fluid from chamber to patient connection point. |
| Luer-type distal connector | Connects to an IV catheter hub, extension set, or needle-free access device per facility protocol. |
Intended Use
This set is intended for administering blood and blood components via gravity flow. It is commonly selected when a dedicated blood administration line is required and when the clinician needs continuous drip visualization and filtration during transfusion. The integrated blood filter supports standard transfusion practice by removing larger particulate matter before it reaches the patient line.
Compatibility and Clinical Considerations
The non-vented design is suited to flexible containers and is not intended to provide air replacement for rigid bottles that require venting for flow. Use only with compatible blood containers and in accordance with institutional transfusion policies, including verification, priming, and monitoring requirements. When priming, facilities typically use an appropriate isotonic solution (often normal saline) per protocol to avoid incompatibility with blood products. The set is designed for single-patient use and should be replaced per facility policy or if occlusion, contamination, or damage is suspected.
Packaging and Handling
Gravity Blood Sets are supplied sterile for immediate clinical use and are intended to be discarded after completion of transfusion or within the timeframe required by local policy. Inspect packaging integrity prior to use, and ensure all connections are secure to support a closed, leak-resistant fluid pathway throughout administration.